Bekslosterid
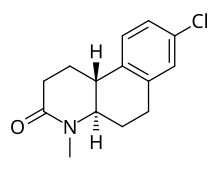
Bekslosterid (LY-191,704) je potentan i nekompetitivan inhibitor enzima 5α-reduktaze. On je srodan sa finasteridom i dutasteridom.[1][2] On je selektivan za izoformu enzima tipa I.[1] Ovaj materijal nije dospeo do tržišta.
 | |
| IUPAC ime | |
|---|---|
(4aS,10bR)-8-hloro-4-metil-1,2,4a,5,6,10b-heksahidrobenzo[f]hinolin | |
| Klinički podaci | |
| Drugs.com | Monografija |
| Identifikatori | |
| CAS broj | 202189-78-4 |
| ATC kod | none |
| PubChem | CID 166562 |
| ChemSpider | 145762 |
| UNII | 36X732P4P0 |
| ChEMBL | CHEMBL24955 |
| Hemijski podaci | |
| Formula | C14H16ClNO |
| Molarna masa | 249,736 |
| |
| |
Sinteza уреди
Osobine уреди
Bekslosterid je organsko jedinjenje, koje sadrži 14 atoma ugljenika i ima molekulsku masu od 249,736 Da.
| Osobina | Vrednost |
|---|---|
| Broj akceptora vodonika | 1 |
| Broj donora vodonika | 0 |
| Broj rotacionih veza | 0 |
| Particioni koeficijent[4] (ALogP) | 2,9 |
| Rastvorljivost[5] (logS, log(mol/L)) | -3,6 |
| Polarna površina[6] (PSA, Å2) | 20,3 |
Reference уреди
- ^ а б Chang, Chawnshang (2002). Androgens and androgen receptor : mechanisms, functions, and clinical application. Boston: Kluwer Academic Publishers. ISBN 1-4020-7188-4.
- ^ Lednicer, Daniel (2008). Strategies for Organic Drug Synthesis and Design. New York: Wiley-Interscience. ISBN 0-470-19039-6.
- ^ Wikel, J. H.; Bemis, K. G.; Audia, J. E.; McQuaid, L. A.; Jones, C. D.; Pennington, P. A.; Lawhorn, D. E.; Hirsch, K. R.; Stamm, N. B. (1993). „QSAR study of benzoquinolinones as inhibitors of human type 1 5-α-reductase”. Bioorganic & Medicinal Chemistry Letters. 3 (6): 1157. doi:10.1016/S0960-894X(00)80306-6.
- ^ Ghose, A.K.; Viswanadhan V.N. & Wendoloski, J.J. (1998). „Prediction of Hydrophobic (Lipophilic) Properties of Small Organic Molecules Using Fragment Methods: An Analysis of AlogP and CLogP Methods”. J. Phys. Chem. A. 102: 3762—3772. doi:10.1021/jp980230o.
- ^ Tetko IV, Tanchuk VY, Kasheva TN, Villa AE (2001). „Estimation of Aqueous Solubility of Chemical Compounds Using E-State Indices”. Chem Inf. Comput. Sci. 41: 1488—1493. PMID 11749573. doi:10.1021/ci000392t.
- ^ Ertl P.; Rohde B.; Selzer P. (2000). „Fast calculation of molecular polar surface area as a sum of fragment based contributions and its application to the prediction of drug transport properties”. J. Med. Chem. 43: 3714—3717. PMID 11020286. doi:10.1021/jm000942e.
Literatura уреди
- Hardman JG, Limbird LE, Gilman AG (2001). Goodman & Gilman's The Pharmacological Basis of Therapeutics (10. изд.). New York: McGraw-Hill. ISBN 0071354697. doi:10.1036/0071422803.
- Thomas L. Lemke; David A. Williams, ур. (2007). Foye's Principles of Medicinal Chemistry (6. изд.). Baltimore: Lippincott Willams & Wilkins. ISBN 0781768799.